
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
This article needs more reliable medical references for verification or relies too heavily on primary sources. (June 2016) |  |
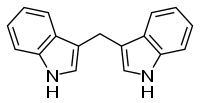
| |
| Names | |
|---|---|
| Preferred IUPAC name
3,3′-Methylenedi(1H-indole) | |
| Other names
3-(1H-Indol-3-ylmethyl)-1H-indole
3,3′-Methylenebis-1H-indole DIM | |
| Identifiers | |
3D model (JSmol)
|
|
| 223072 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.124.716 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C17H14N2 | |
| Molar mass | 246.313 g·mol−1 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335, H413 | |
| P261, P264, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
3,3′-Diindolylmethane (DIM) is a compound derived from the digestion of indole-3-carbinol, found in cruciferous vegetables, such as broccoli, Brussels sprouts, cabbage and kale.[1] It and its parent compound – indole-3-carbinol – are under laboratory research to determine their possible biological properties, particularly in anti-cancer mechanisms.[1][2][3] DIM is sold as a dietary supplement.[1]
Properties
In vitro, DIM has action as a histone deacetylase inhibitor,[1] specifically against HDAC1, HDAC2, and HDAC3.[4] DIM is a metabolite of indole-3-carbinol.[1][2][3] DIM was found to be a mild cannabinoid agonist with low binding affinity for both CB1 and CB2.[5][6]
Researchers from Ben-Gurion University of the Negev, in collaboration with teams from Sichuan University and the National University of Singapore, have identified that 3,3′-Diindolylmethane (DIM) – a naturally occurring molecule also referred to as bisindole – can reduce biofilms responsible for plaque and cavities by a remarkable 90%. [7]
See also
- Glucobrassicin, precursor to indole-3-carbinol
- Phytochemicals
References
- ^ a b c d e "Indole-3-carbinol". Micronutrient Information Center, Linus Pauling Institute, Oregon State University. 1 July 2017. Retrieved 11 July 2022.
- ^ a b "Indole-3-Carbinol". Memorial Sloan Kettering Cancer Center. Retrieved 2019-09-03.
- ^ a b "3,3'-Diindolylmethane". PubChem, US National Library of Medicine. 9 July 2022. Retrieved 11 July 2022.
- ^ Rajendran, P; Ho, E; Williams, DE; Dashwood, RH (2011). "Dietary phytochemicals, HDAC inhibition, and DNA damage/repair defects in cancer cells". Clinical Epigenetics. 3 (1): 4. doi:10.1186/1868-7083-3-4. PMC 3255482. PMID 22247744.
- ^ Andhika B. Mahardhika; et al. (March 2023). "Design, synthesis, and structure–activity relationships of diindolylmethane derivatives as cannabinoid CB2 receptor agonists". Archiv der Pharmazie. 356 (3): e2200493. doi:10.1002/ardp.202200493. PMID 36437108.
- ^ Paolo Tucci; et al. (2023). "The Plant Derived 3-3′-Diindolylmethane (DIM) Behaves as CB2 Receptor Agonist in Prostate Cancer Cellular Models". International Journal of Molecular Sciences. 24 (4): 3620. doi:10.3390/ijms24043620. hdl:2164/20232. PMC 9962283. PMID 36835033.
- ^ Yifat Baruch; et al. (6 June 2023). "3,3′-Diindolylmethane (DIM): A Potential Therapeutic Agent against Cariogenic Streptococcus mutans Biofilm". Antibiotics. 12 (6): 1017. doi:10.3390/antibiotics12061017. PMC 10295630. PMID 37370336.
External links
- Diindolylmethane Information Resource Center, University of California-Berkeley
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
